|
The SI unit of pressure is Pascal, which is equal to Newton per square meter (N/m²). 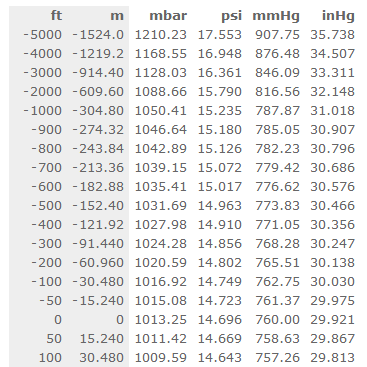
Pressure scalar quantity, because it has value but has no direction. Again, more pressure is required in the area. This means that if we apply a force in a single case, it will be a force.Īs an example, it is easy to take a photo with a pin because the area is less. On the other hand, the force exerted on a single area is called pressure. 
In the language of physics, a force is an object that changes or tries to change its state by acting on it or that changes or tries to change its motion by acting on a moving object.įor example, if someone pushes against a wall, the force is applied whether it changes its position or not. This unit is used extensively in the measurement of blood pressure and barometric pressure. It is defined as the amount of pressure exerted by a column of mercury that is 1 millimeter high and has a density of 13.5951 g/cm³. In summary, millimeters of mercury (mmHg) is a unit of pressure measurement that is commonly used in medicine and meteorology. To convert mmHg to kPa, the following equation is used: 1 mmHg = 0.133 kPa. Barometric pressure is an important factor in weather forecasting as it helps predict weather patterns and changes.Īnother unit of pressure measurement that is commonly used is the kilopascal (kPa). In addition to medicine, mmHg is also commonly used in meteorology to measure barometric pressure, which is the pressure exerted by the Earth's atmosphere. This means that the pressure exerted by the blood in the arteries when the heart beats (systolic pressure) is around 120 mmHg, and the pressure exerted when the heart is at rest (diastolic pressure) is around 80 mmHg. The normal range for blood pressure in adults is around 120/80 mmHg. In the case of mmHg, it is the amount of force exerted by a column of mercury that is 1 millimeter high. Pressure is the amount of force exerted per unit area. To understand the concept of mmHg, it is important to first understand what pressure is. This unit of pressure is used extensively in medicine, especially in the measurement of blood pressure. The mmHg unit is defined as the amount of pressure exerted by a column of mercury that is 1 millimeter high and has a density of 13.5951 g/cm³, which is the density of mercury at 0☌. It is also known as millimetres of water column, which is abbreviated as mmWC. Type in unit symbols, abbreviations, or full names for units of length, area, mass, pressure, and other types.Millimeters of mercury (mmHg) is a unit of pressure measurement that is commonly used in medicine and meteorology. You can find metric conversion tables for SI units, as well as English units, currency, and other data. It is approximately equal to Earth's atmospheric pressure at sea level.Ĭ provides an online conversion calculator for all types of measurement units. It is sometimes used as a reference pressure or standard pressure. The standard atmosphere (symbol: atm) is a unit of pressure defined as 101325 Pa (1.01325 bar). This small difference is negligible for most applications outside metrology. The difference between one millimeter of mercury and one torr, as well as between one atmosphere (101.325 kPa) and 760 mmHg (101.3250144354 kPa), is less than one part in seven million (or less than 0.000015%). The relationship between the torr and the millimeter of mercury is: The decimal form of this fraction is approximately 133.322368421. Therefore, 1 Torr is equal toġ01325/760 Pa. The torr is defined as 1/760 of one standard atmosphere, while the atmosphere is defined as 101325 pascals. The millimeter of mercury by definition is 133.322387415 Pa (13.5951 g/cm3 × 9.80665 m/s2 × 1 mm), which is approximated with known accuracies of density of mercury and standard gravity.  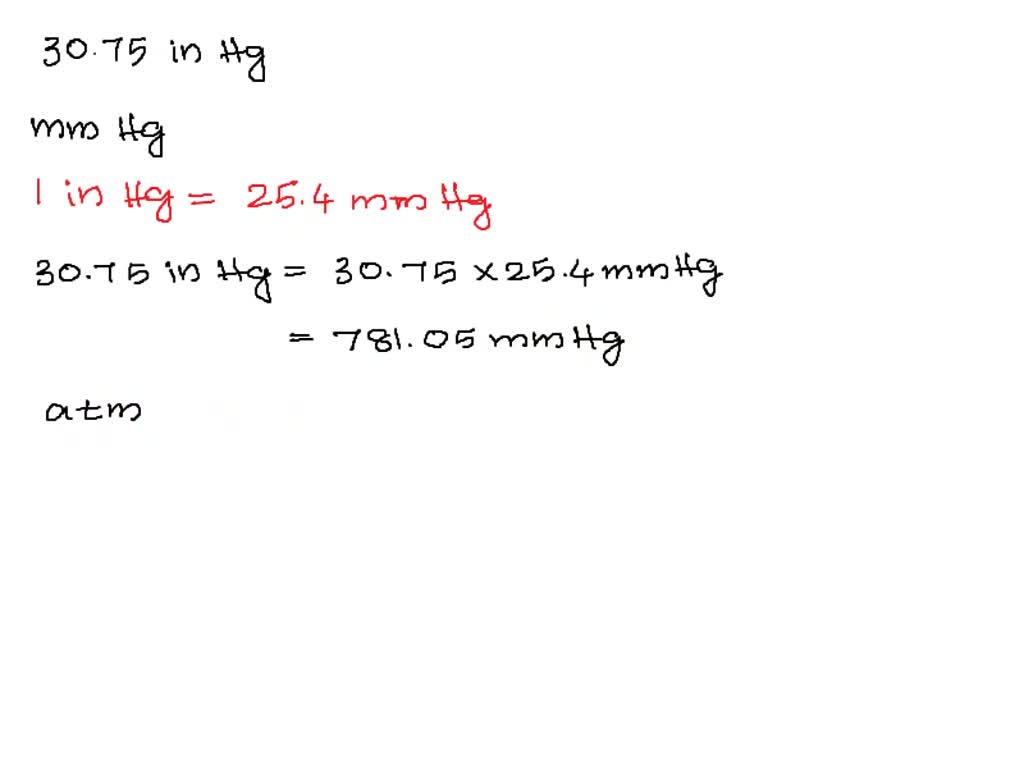
You can do the reverse unit conversion fromĪtmosphere to mm Hg, or enter any two units below: Enter two units to convert From:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
